Abstract
SUMMARY: The acetazolamide (ACZ) challenge test is a useful clinical tool and a reliable predictor of critically reduced perfusion. In patients with chronic steno-occlusive disease, the ability to maintain normal cerebral blood flow by reducing vascular resistance secondary to autoregulatory vasodilation is compromised. Identification of the presence and degree of autoregulatory vasodilation (reflecting the cerebrovascular reserve) is a significant prognostic factor in patients with chronic cerebrovascular disease. The pharmacologic challenge of a vasodilatory stimulus such as ACZ can also be used to optimize the treatment strategies for these patients. The pathophysiology, methods, and clinical applications of the ACZ challenge test are discussed in this article.
Viability of the cerebral parenchyma is dependent on the ability of the brain vasculature to provide adequate levels of cerebral blood flow (CBF). In patients with chronic steno-occlusive disease, the ability to maintain normal CBF by reducing vascular resistance is compromised. Identification of the degree of autoregulatory vasodilation reflects cerebrovascular reserve (CVR), which is a significant prognostic factor in chronic cerebrovascular disease.1–5 Flow reserve can be assessed with the use of paired blood flow measurements, with the initial measurement obtained at baseline and the second, after a vasodilatory stimulus, such as acetazolamide (ACZ).6
Pathophysiology of Chronic Cerebrovascular Disease
Chronic cerebral hypoperfusion is usually the result of occlusion or stenosis of large arteries in the neck or the circle of Willis. Clinical symptoms and manifestations of brain ischemia in patients with chronic cerebrovascular disease (CVD) develop as a consequence of 2 main mechanisms: embolic events from atherosclerotic plaques resulting in local compromise of blood flow and systemic hemodynamic alterations that further reduce an already compromised cerebral perfusion state.7
The hemodynamic changes due to a decline in cerebral perfusion pressure have been studied by many investigators.6,8–11 Chronic CBF restriction causes a progressive decrease in cerebral perfusion pressure (CPP). Initially, decreases of CPP cause varying degrees of autoregulatory vasodilation of small distal arterioles.12 Powers10 and Powers et al11 proposed a 2-stage classification of hemodynamic impairment in patients with CVD. In stage I (autoregulatory vasodilation), autoregulation reduces cerebral vascular resistance. CBF and oxygen extraction fraction (OEF) are not significantly changed. Increases of cerebral blood volume (CBV) and mean transit time (MTT) are 2 parameters that reflect this initial phase of compensatory autoregulatory vasodilation. Further decreases of CPP beyond cerebral autoregulatory vasodilation capacity eventually result in stage II (autoregulatory failure), characterized by decreases of CBF and increases of OEF. When the CBF decreases, neurons increase the fraction of oxygen extracted from the blood to maintain normal neurologic function.13 This phenomenon of reduced CBF and increased OEF has been termed “misery perfusion.”11,14
Derdeyn et al9 have more recently shown that autoregulatory vasodilation and increased oxygen extraction occur simultaneously. Slight reductions in CBF through the autoregulatory range lead to slight but measurable increases in OEF.15 When autoregulatory capacity is finally exceeded, CBF decreases more rapidly and OEF increases dramatically.16 The CBV is more variable in the autoregulatory range.9 The cerebral rate for oxygen metabolism (CMRO2) remains unchanged over the initial and late decreased levels of CPP.9,17 As the CPP falls further, oxygen extraction reaches a maximum. Further decreases in CPP will result in an inability to maintain adequate blood flow and lead to varying degrees of brain ischemia.
Evaluation of these compensatory mechanisms is important in patients with CVD to determine the risk of future ischemic events and in the selection and planning of therapeutic interventions. Generally speaking, 3 approaches can be used in the evaluation of patients with CVD6: One requires the measurement of the CBV/CBF ratio, mathematically equivalent to the MTT. The second approach (and the focus of this article) attempts to determine the degree of cerebral flow reserve. This is accomplished by comparing CBF under baseline conditions and after a vasodilatory stimulus such as ACZ. Finally, direct measurements of OEF to identify patients with increased oxygen extraction (currently performed by using positron-emission tomography [PET]) can be performed.
Cerebrovascular Reactivity
Alterations in blood flow secondary to a vasodilatory stimulus (such as ACZ) can be used to estimate CVR, which is calculated as the percentage increase in CBF after ACZ relative to baseline1,18: 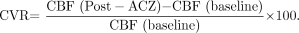 Vascular territories harboring vaso-occlusive disease undergo compensatory vasodilation up to a maximal level. This precludes further dilation of the arterioles in the affected region in response to ACZ; therefore, the expected normal increases of CBF following ACZ challenge are blunted compared with normal brain parenchyma.
Vascular territories harboring vaso-occlusive disease undergo compensatory vasodilation up to a maximal level. This precludes further dilation of the arterioles in the affected region in response to ACZ; therefore, the expected normal increases of CBF following ACZ challenge are blunted compared with normal brain parenchyma.
On the basis of studies using stable xenon-enhanced CT (Xe-CT) and ACZ challenge, Rogg et al19 classified 3 types of patient responses to ACZ:
Type I patients have normal baseline CBF that increases after ACZ challenge.
Type II patients have areas of decreased CBF on baseline studies that increase after ACZ administration.
Type III patients have decreased CBF at baseline and a paradoxic continued reduction in regional CBF after ACZ administration.
Type III responses appear to define those patients who are the most likely to benefit from surgical revascularization.19 This final category likely relates to a steal phenomenon, indicating a decrease in blood flow in regions that are already maximally dilated,20,21 identified with quantitative CBF measurements.22
ACZ
ACZ is a carbonic anhydrase inhibitor that penetrates the blood-brain barrier slowly and acts as a cerebral vasodilator agent. Inhibition of carbonic anhydrase causes carbonic acidosis, which induces a considerable increase in CBF.23 ACZ is safe to administer and is generally well tolerated. Systemic blood pressure, heart and respiratory rates, arterial pH, arterial CO2 pressure, and CMRO2 are unaffected. Most common side effects are acute and transient, including transient circumoral numbness, paresthesias, malaise, and headache.23,24 Reversible pontine ischemia caused by ACZ challenge has been discussed in a case report25; however, Piepgras et al26 reported no acute ischemic sequelae in more than 1000 studies with the use of ACZ.
Contraindications to ACZ administration are hypersensitivity to other sulfonamides, electrolyte disturbances, marked kidney and liver disease, adrenocortical insufficiency, and long-term use in chronic noncongestive angle-closure glaucoma.27
A standard dose of 1000 mg intravenously is used for the ACZ challenge test. Peak CBF augmentation occurs at approximately 10–15 minutes after intravenous bolus administration. A 30%–60% increase in CBF is achieved in healthy subjects.28 Criteria that have been used to define an abnormal response to ACZ include <10% increase in the absolute CBF or an absolute change of <10 mL/100 g/min.29
Imaging Techniques
There is a large armamentarium of imaging techniques for assessing the adequacy of cerebral perfusion. These include PET, single-photon emission CT (SPECT), Xe-CT, dynamic perfusion CT (PCT), MR imaging dynamic susceptibility contrast, arterial spin-labeling (ASL), and transcranial Doppler sonography.
Xe-CT
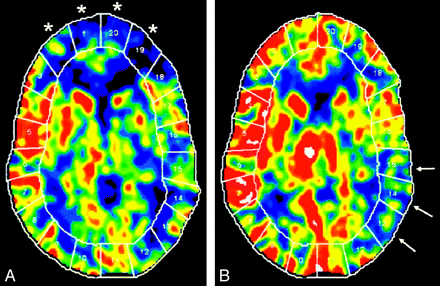
Xe-CT combined with ACZ challenge is an established technique, which has been used for >20 years to evaluate chronic cerebral ischemia. It provides quantitative assessment of CBF and CVR (Fig 1). The inhaled xenon gas dissolves in blood rapidly and freely crosses the blood-brain barrier and concentrates in the brain by virtue of its liposolubility. CBF is calculated by measuring the rate of xenon clearance from the brain by using the Kety-Schmidt model, which provides reliable CBF quantization, along with high-resolution imaging.28,30 Because of the short cerebral residence time of inhaled xenon, the study can be repeated in the same session. Xe-CT is an expensive and complex examination, requiring excellent patient cooperation, the presence of an anesthetist, and the use of specialized and expensive equipment. Side effects of xenon inhalation, such as a decrease in respiratory rate, headache, nausea, vomiting, and convulsions may occasionally occur.31 In the United States, xenon is not currently approved by the US Food and Drug administration for clinical Xe-CT assessment.
Xe-CT CBF maps in a patient with Moyamoya disease. A, Baseline. B, After ACZ administration. Baseline scan (A) shows reduced CBF in the bilateral ACA and anterior watershed areas (areas 1, 2, 19, and 20, asterisk). After ACZ, there is a robust increase in the CBF, indicating a normal cerebral reserve in these territories. There is reduced baseline flow with decreased augmentation of CBF after ACZ, indicating poor cerebral reserve in the left posterior MCA and the left posterior watershed territories (areas 13–15, arrows).
PCT
PCT is a noninvasive method that provides CBF, CBV, and MTT values and can be combined successfully with ACZ to assess cerebral hemodynamics more completely. The feasibility of CTP in the evaluation of chronic ischemia has been encouraging in various studies.32–34 Furukawa et al35 studied the usefulness of PCT in the evaluation of chronic cerebral ischemia and compared the technique with Xe-CT. In their study, the relative scores of CBF obtained by PCT correlated well with Xe-CT values, though the absolute values did not show as good a correlation. CTP imaging has also been validated against PET.36 Bisdas et al37 studied 12 patients with chronic carotid stenosis who underwent dynamic PCT and PET studies.37 CBF measurements from PCT correlated well with PET-derived CBF values. This study also reported overestimation of CBF in PCT compared with PET after ACZ challenge. This has also been reported by Kudo et al,38 who hypothesized that inclusion of surface blood vessels and perforating arteries very likely leads to the overestimation by PCT.38 Vascular pixel elimination in PCT analysis can minimize this problem.38
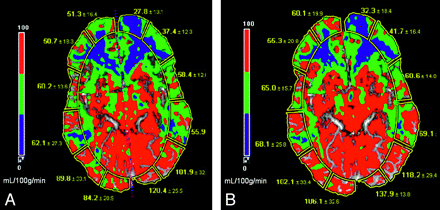
PCT with ACZ challenge relies on quantitative assessment; therefore, the reproducibility of the perfusion parameters is important. Establishing a uniform and standard postprocessing technique is essential for maintaining good reproducibility (Fig 2).39,40 Use of standard head immobilization techniques and adequate patient coaching are important to minimize patient motion. Waaijer et al41 studied PCT images in 20 patients with unilateral symptomatic carotid artery stenosis to assess the reproducibility of quantitative CTP parameters. This study revealed that MTT is the most reproducible parameter for regional measurements of PCT and that the use of CBV and CBF ratios results in better reproducibility compared with absolute CBV and CBF values for this patient group.41
A 34-year-old man with severe headache and blurry vision was diagnosed with Moyamoya disease. CT perfusion maps. A, Baseline. B, After ACZ administration. CBF (measured in mL/100 g/min) is diminished in bilateral ACA, ACA-MCA watershed, and the MCA territories (depicted as blue to green). These areas show a very suboptimal increase in CBF after ACZ administration and thus exhibit limited CVR. Note the increased CBF secondary to vasodilatory capacity in the PCA and MCA-PCA distribution after ACZ administration.
The choice of a reference artery is critical for accurate CBF measurements in CTP, both in acute and chronic ischemia. However, no general consensus has been reached regarding whether the arterial input function (AIF) ipsilateral or contralateral to the side of stenosis is more accurate.37,42 Moreover, the choice of the AIF may be more complex in chronic carotid occlusion because collateral formation through the circle of Willis results in delay and dispersion of the contrast bolus. In a recent study comparing PCT with PET in patients with chronic cervical carotid artery occlusion, Kamath et al43suggested that CBF values obtained by PCT and PET compared favorably when processed by using a dedicated AIF for each territory.43 In Moyamoya disease, both anterior and middle cerebral arteries (ACA, MCA) may be occluded. In these cases, the basilar artery or the P1 segment of the posterior cerebral artery (PCA) should be selected.44
The advantages of PCT are that it is a rapid, noninvasive, and readily available method that provides CBF, CBV, and MTT values and can be combined successfully with ACZ to assess cerebral hemodynamics fully. Also, anatomic vascular imaging such as CT angiography can be performed at the same time. One current drawback of PCT is restricted spatial coverage. Continually improving CT technology (wider detector arrays and controlled table movements) will very likely overcome this problem. Other inherent disadvantages of PCT include the risks of ionizing radiation and iodinated contrast. Further studies are needed to establish the accuracy, reliability, and reproducibility of PCT-derived quantitative measurements, but the present data regarding CVR assessment are encouraging.
MR Perfusion
Dynamic Contrast Bolus MR Perfusion
MR imaging can detect the changes in magnetic susceptibility during passage of a compact bolus injection of contrast and can yield relative and absolute hemodynamic values of brain perfusion. Dynamic susceptibility contrast MR perfusion, similar to CTP, has the advantages of widespread availability, high spatial resolution, and the ability to obtain concomitant anatomic vascular mapping. As an added advantage, there is a more complete coverage of the brain and no ionizing radiation. One of the disadvantages of MR perfusion is the complex relation between the signal intensity and contrast concentration, resulting in difficulties with absolute perfusion parameter calculation.45,46
Assessment of CVR with the ACZ challenge has been described with MR perfusion studies.47–51 Perfusion MR imaging before and after ACZ administration compares favorably with technetium Tc99m ethyl cysteinate dimmer (Tc99m-ECD) SPECT for the detection of impaired CVR.52 Agreement between PET and MR perfusion was moderate for physiologic CBF values at rest and after the ACZ challenge in healthy humans.53 Similar to CTP, the CBF values reported with MR perfusion were overestimated compared with the gold standard of PET.54–56
ASL MR Perfusion
ASL is a MR perfusion technique that takes advantage of arterial water as a freely diffusible tracer to measure CBF. Because exogenous contrast is not required for this technique, it is completely noninvasive and repeatable. There are only a limited number of studies in humans assessing the feasibility of ASL in chronic ischemia. The combination of ACZ challenge and ASL MR perfusion in CVD produced the expected results of CBF alteration.51 Obtaining quantitative accurate CBF data is challenging; however, test-retest reproducibility of these techniques has been encouraging.50 A study of a small number of patients with chronic arterial stenosis compared ASL perfusion with ACZ challenge with iodine 123 N-isopropyl-p-iodoamphetamine (123I-IMP) SPECT and demonstrated concordant results.57 A recent study of a large number of patients with ASL suggested the potential use of this technique for measuring CVR and for serial assessment following revascularization.58,59
PET
Physiologic evaluation of chronic CVD can also be performed with PET, which measures the CBF and OEF to estimate cerebral perfusion. It offers the advantage of providing quantitative evaluations useful for the assessment of various parameters of brain metabolism and physiology. PET techniques are not as readily available because the radioisotopes have very short half-lives and need to be produced by cyclotrons, a technology only available in large medical centers or in central commercial radiopharmacies.
Several studies using PET have been conducted to correlate various pathophysiologic aspects of early ischemia with the final outcome of brain tissue at risk for infarction. Patients classified as having hemodynamic ischemia were found in 1 study to have low CBF associated with decreases of CMRO2 on the side of the arterial occlusion.60 PET measurement of increased OEF has been shown to be an important and independent predictor of subsequent stroke. 61,62 Generally speaking, an increased OEF should correspond to decrease in CVR when assessed by challenge tests. These 2 measures, though strongly related, are not equivalent. Recent studies demonstrated that decreases in CVR and increases in OEF did not necessarily parallel each other.63–65
PET measurements have been useful in demonstrating reversals of altered OEF because patients improve brain flow through collaterals66 and following superficial temporal-to-MCA anastomoses.67,14 Controlled studies to assess the value of OEF determinations for selection of therapy in patients with carotid occlusive disease are needed to determine the utility of these techniques.
SPECT
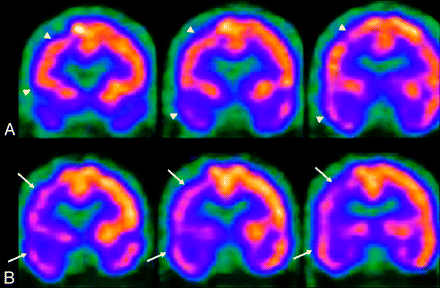
Currently, SPECT is the most readily available nuclear medicine technique for assessment of cerebral hemodynamics and uses radionuclides that concentrate in neurons in direct relation to flow. Technetium Tc99m hexamethylpropyleneamine oxime (HMPAO) and Tc99m-ECD are 2 Technetium Tc99m compounds used in routine clinical practice. There are significant differences in the pharmacokinetics and brain distribution between these 2 agents, but in general, they are equivalent for imaging of the brain. SPECT images with these 2 radionuclides represent the distribution and concentration of the radionuclide within the brain parenchyma according to blood flow and are mainly evaluated qualitatively by visual inspection (Fig 3). Some semiquantitative techniques exist that provide a more objective assessment, especially useful when combined with ACZ challenge. However, currently there are no practical absolute quantitative techniques to measure CBF by using SPECT.
SPECT perfusion study of a 64-year-old man with high-grade right M1 and A1 segment stenosis. A, Baseline. B, After ACZ administration. There is decreased uptake and perfusion (arrows) involving the right frontal, parietal, and temporal lobes on the ACZ study (B), which resolve during baseline conditions (arrowheads).
Brain SPECT has also been validated in comparison studies with O-15 H2O PET for evaluation of cerebrovascular reactivity to ACZ challenge in patients with CVD.68,69 Several studies have demonstrated the usefulness of brain SPECT in conjunction with ACZ challenge for evaluation of vascular reactivity in patients with various types of vaso-occlusive disease.70–72 Responses to ACZ have been studied in patients with Moyamoya disease.73 Brain SPECT with Tc99m HMPAO and ACZ challenge was used successfully in the assessment of brain hemodynamics in 15 patients with CVD before and after bypass surgery.74
Although evaluation of cerebral hemodynamics in patients with CVD is best performed by using PET, SPECT imaging with ACZ challenge offers a more practical alternative for many centers.
Clinical Applications of the ACZ Challenge
Stroke Risk Assessment
Identification of compromise in CVR capacity is important in the evaluation of ischemic stroke. The degree of vascular occlusion in this situation is an incomplete indicator of future stroke risk.75 There may be a synergistic effect between embolic phenomenon from atherosclerotic plaque and impaired hemodynamics causing ischemic stroke in large artery atherosclerotic occlusions.76,77 Compromised CVR is a key determinant of ischemic infarction, regardless of whether it is precipitated by embolic or hemodynamic factors.75
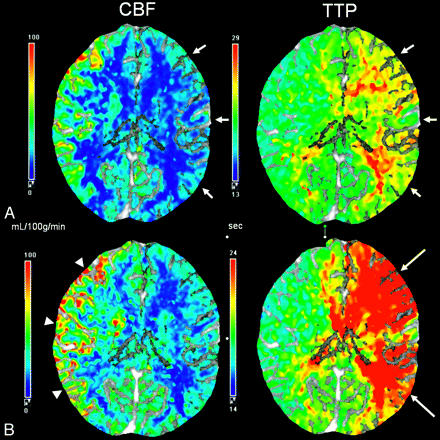
Kuroda et al2 describe a prospectively studied longitudinal cohort of 77 patients in which patients with decreased CBF and CVR resulting from internal carotid artery (ICA) or MCA occlusion had a higher risk of new ischemic stroke than those without. This was one of the first prospective studies to confirm the value of CVR as a predictor of the future stroke risk.2 Another prospective study demonstrated that decreased cerebrovascular reactivity to ACZ (determined quantitatively by 133Xe SPECT) is an independent predictor of the 5-year risk of subsequent stroke in patients with symptomatic major cerebral artery occlusion.5 Multiple studies of patients with carotid occlusive disease have consistently shown a higher incidence of ischemic complications in those patients with compromised vascular reserve (Fig 4).3,4,62,78
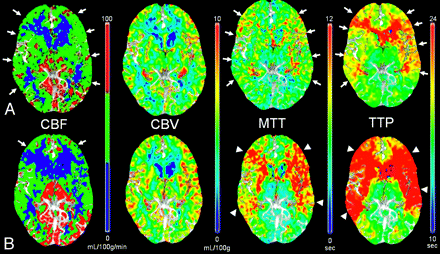
CTP. Chronic left internal carotid artery occlusion. A, Baseline. B, After ACZ administration. There is significant hypoperfusion in the left hemisphere at baseline with decreased CBF and increased TTP (A, short arrows). After administration of ACZ (B), CBF decreases throughout the left hemisphere, resulting in negative calculated CVR percentages. The left-sided TTP increases even further in post-ACZ flow maps as seen by the accentuated asymmetry (B, long arrows). Note the normal increase in the right-sided CBF after the vasodilatory stimulus of ACZ (B, arrowheads).
ACZ-defined CVR can also be used as an additional assessment tool in asymptomatic carotid artery stenosis.7,79 In a prospective study of 94 patients with asymptomatic carotid artery stenosis of >70%, Silvestrini et al79 suggested a link between impaired cerebrovascular reactivity and the risk of ischemic events ipsilateral to severe asymptomatic carotid stenosis.
The risk of perioperative cerebral infarction during carotid and cardiac surgery can be estimated by using CVR. Schoof et al80 prospectively studied 2797 patients with carotid artery stenosis/occlusion undergoing cardiac surgery with cardiopulmonary bypass and assessed cerebral autoregulation by using transcranial Doppler sonography with CO2 stimulation. Increased risk of perioperative stroke was observed in patients with high-grade stenosis or occlusion and exhausted autoregulatory reserve, suggesting that assessment of CVR facilitates identification of patients with an excess perioperative stroke risk.80
Moyamoya Disease
Moyamoya disease is a well-described entity characterized by progressive stenosis and occlusion of the supraclinoid ICA and its branches, affecting both the pediatric and adult population and frequently resulting in cerebral infarctions.81 Medical therapy for Moyamoya disease is ineffective; consequently, direct and indirect surgical revascularization has become the primary technique of treatment for these patients to reduce the incidence of ischemic sequelae. Many series have reported the efficacy of revascularization in this syndrome by using primarily direct superficial temporal artery (STA)-MCA bypass.82–89
PET studies in Moyamoya disease have demonstrated reduced CBF and CO2 response and increased CBV, MTT, and OEF.90 CBF is significantly lower in patients with Moyamoya disease than in healthy subjects of the same age. Also the CBF has a dominant posterior distribution in contrast to the anterior distribution observed in healthy control subjects (Fig 5).91
CT perfusion maps in a 51-year-old patient presenting with right-sided hemiparesis who was diagnosed with Moyamoya disease, demonstrating bilateral supraclinoid internal carotid occlusion. A, Baseline. B, After ACZ administration. The baseline pre-ACZ PCT (A) demonstrates the typical pattern of Moyamoya disease with decreased CBF and increased MTT and TTP in the bilateral anterior and middle cerebral distributions (arrows). After ACZ challenge (B), the CBF in the anterior circulation decreases consistent with steal phenomenon (B, CBF, arrows). The CBF map demonstrates a normal expected increase in the PCA territories. There is further prolongation of the MTT and TTP in both ACA and MCA distributions (arrowheads, B), consistent with worsening of cerebral hemodynamics after ACZ, and type III physiology. The patient successfully underwent left-sided ECIC bypass surgery.
CVR testing with ACZ challenge is useful in Moyamoya disease. CVR in the anterior cerebral and middle cerebral territories is significantly lower than that in the posterior cerebral territory and the central region around the basal ganglia.92 Of multiple hemodynamic parameters measured by ACZ challenge PCT in adult Moyamoya disease, percentage change in CBF correlated most significantly with the angiographic stage of the disease.93 Another study correlated ACZ-challenged PCT with SPECT in patients with Moyamoya disease and showed that baseline PCT parameters, including CBV and MTT, correlated weakly with the CVR obtained with SPECT.33 The study demonstrated that percentage change of CBF correlated well with quantitative SPECT CVR measurements.33
Pre- and Postoperative Evaluation of Extracranial-Intracranial Bypass for Flow Augmentation
Cerebral revascularization by using extracranial−intracranial (ECIC) bypass is a treatment option in the setting of major cerebral artery occlusive disease. A large ECIC bypass study was initiated to determine whether anastomosis of the STA to the MCA could reduce ischemic stroke and stroke-related death among patients with symptomatic surgically inaccessible (to carotid endarterectomy [CEA]) atherosclerotic stenosis or occlusion of the ICA or MCA.94 Although this study failed to show the efficacy of bypass over medical management for anterior circulation occlusive disease, it preceded effective noninvasive tools for CBF testing. With greater understanding of the importance of assessing cerebral hemodynamics in patients with CVD, interest in revascularization has re-emerged.
In recent years, the use of ECIC bypass for anterior circulation ischemia in selected patients has been reported in several studies.95–97 Several case series have demonstrated benefit from ECIC bypass if the patient population is carefully defined. In a study by Mendelowitsch et al,98 85% of the patients had no further cerebrovascular events after surgery in the average follow-up period of 44 months, and only 11% experienced another cerebrovascular event. Following ECIC bypass, most (95.4%) patients experience cessation of their ischemic events and stabilization of pre-existing neurologic dysfunction.99 To assess the efficacy of ECIC bypass in this group more definitively, a randomized trial, the Carotid Occlusion Surgery Study, is underway in North America.100 This study is funded by the National Institutes of Health, which stratifies patients as candidates for surgery only if they manifest increased OEF on PET.
Hemodynamic assessment, including CVR testing, represents an important assessment tool after ECIC bypass surgery. After ECIC bypass, several studies have shown that there can be full or partial reversal of impaired CVR (Fig 6).21,101,102 One study using 133Xe SPECT in 28 patients who underwent ECIC bypass showed significant improvement of CVR after surgery while the resting CBF was essentially unchanged.103 Schaller104 concluded that the hemodynamic parameters observed in patients who experience improved neurologic function or diminished stroke risk profile after ECIC-bypass surgery contain both significantly elevated OEF and CBF/CBV.
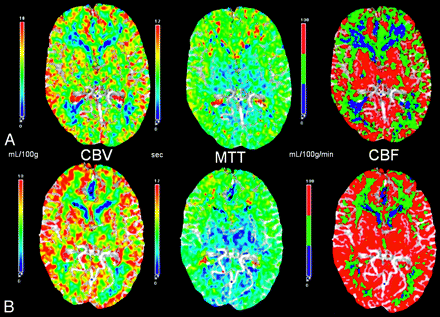
CT perfusion flow maps in a 56-year-old patient presenting with multiple transient ischemic episodes and diagnosed with Moyamoya disease with bilateral carotid occlusion on digital subtraction angiography before (A) and 12 months after (B) bilateral ECIC bypass surgery. The preoperative baseline maps (A) show a typical pattern of Moyamoya disease with decreased CBF and increased MTT in the anterior circulation. After bilateral ECIC bypass (STA-MCA, B), there is an increase in the CBF in the ACA and MCA territories with minimal decrease in the MTT. The improvement is more pronounced in the MCA territory because of the proximity of the graft. Quantitatively, there is improvement in the CVR in these distributions. After bypass, the patient's ischemic symptoms resolved.
Although the role of cerebral revascularization in patients with major cerebral artery occlusive diseases remains controversial, there is growing evidence that symptomatic patients with a documented hemodynamic source of their symptoms are very good candidates for undergoing STA-MCA bypass.
Carotid Balloon Occlusion
Balloon test occlusion of the ICA is performed routinely to assess the collateral circulation before surgical or endovascular procedures that may involve sacrificing or prolonged occlusion of the ICA. Patients who develop any change in the neurologic status during balloon occlusion are thought to demonstrate poor autoregulatory potential. Up to 10% of patients in whom the test is clinically successful may still have diminished CBF in the ipsilateral hemisphere.105,106
Perfusion imaging, pharmacologically induced hypotension, and stump pressure measurements are various methods used for evaluation during the balloon test occlusion.28 The feasibility of the ACZ challenge test during carotid balloon occlusion has been shown in a few studies.105,108 Using ACZ challenge and perfusion CT in a study of 8 patients, Jain et al107 suggested that patients with symmetric CBF and normal vasodilatory capacity would do well after permanent carotid occlusion and that patients with asymmetric CBF and abnormal response to the ACZ challenge test may require a revascularization procedure to protect them from future ischemic sequelae.
Hyperperfusion Syndrome
Cerebral hyperperfusion syndrome is an uncommon but serious complication of carotid revascularization, including CEA and carotid stent placement. Hyperperfusion syndrome (associated with increased CBF compared with preoperative values) results in headache, focal seizure activity, cerebral edema, and intracerebral hemorrhage and is associated with significant mortality and morbidity.29
Komoribayashi et al109 reported hyperperfusion immediately after CEA in 56% of patients with reduced preoperative CVR. Another study measured concentrations of malondialdehyde-modified low-attenuation lipoprotein (MDA-LDL), a biochemical marker of oxidative damage, in serum samples and CVR measurements in 90 patients undergoing CEA.110 This study suggested that reduced preoperative CVR (<20%) and increased MDA-LDL correlate with the development of cerebral hyperperfusion after CEA.110 In a more recent study, measurements of preoperative CBV by perfusion-weighted MR imaging helped to identify patients at risk for cerebral hyperperfusion after CEA.111 Other studies have also shown that preoperative measurement of reduced CVR (<10% CBF response to ACZ) results in the timely and reliable identification of patients at risk for hyperperfusion syndrome.112,113 In these patients, careful monitoring and control of blood pressure should be initiated even intraoperatively.112
Conclusions
In patients with chronic steno-occlusive disease, it is important to assess the capacity of the cerebral circulation to adjust to superimposed hemodynamic changes. Current research suggests that compromised CVR is an important risk factor of future ischemic sequelae. Administration of a vasodilator such as ACZ can assess cerebral reserve by observing cerebrovascular reactivity to hemodynamic stress. The ACZ challenge test is a useful clinical tool and can be used to optimize the treatment strategies for patients with chronic cerebral ischemic disease.
Acknowledgments
We thank Rhonda Strunk for technical assistance.
References
- Received January 4, 2009.
- Accepted after revision January 5, 2009.
- Copyright © American Society of Neuroradiology