Abstract
SUMMARY: Epstein-Barr virus is a ubiquitous herpesvirus that may cause both infective (encephalitis, meningitis, and so forth) and postinfection inflammatory (such as Guillain-Barré syndrome, acute disseminated encephalomyelitis) manifestations in the CNS. Diagnosis of Epstein-Barr virus–related CNS pathologies is often complicated due to a nonspecific clinical presentation and overlap with other infectious and noninfectious causes, both clinically and on imaging. The Epstein-Barr virus is also implicated in several lymphoproliferative disorders in both immunocompromised and immunocompetent hosts. MR imaging is preferred for evaluating the extent of involvement and monitoring therapy response, given its high sensitivity and specificity, though imaging findings may be nonspecific. Herein, we review the imaging spectrum of Epstein-Barr virus–associated CNS disorders.
ABBREVIATIONS:
- ADEM
- acute disseminated encephalomyelitis
- ATM
- acute transverse myelitis
- EBV
- Epstein-Barr virus
- EBNA
- Epstein-Barr nuclear antigen
- Ig
- immunoglobulin
- LG
- lymphomatoid granulomatosis
- LPD
- lymphoproliferative disorder
- MOG
- myelin oligodendrocyte glycoprotein
- PCNSL
- primary CNS lymphoma
- PCR
- polymerase chain reaction
- PTLD
- posttransplant lymphoproliferative disorder
- SMT
- smooth-muscle tumor
The Epstein–Barr virus (EBV), also known as herpesvirus 4 human, belongs to the herpesvirus family. It is highly prevalent and affects >90% of individuals worldwide.1 Primary EBV infection in childhood is usually asymptomatic. It causes infectious mononucleosis in young adults and presents with fever, lymphadenopathy, and tonsillopharyngitis.2 Depending on age and immune status, CNS manifestations may occur in about 18% of patients. These include encephalitis, meningitis, cerebellitis, cranial nerve palsy, Guillain–Barré syndrome, acute disseminated encephalomyelitis (ADEM), and infectious or acute transverse myelitis (ATM).3 Cheng et al4 reported a cohort of 89 children with EBV-related neurologic involvement. Encephalitis and meningoencephalitis (72%) were the most common presentations, followed by Guillain-Barré syndrome (17%), ADEM (3.4%), acute myelitis (2.2%), and posttrantplant lymphoproliferative disorders (PTLDs) (1.1%). EBV also plays a vital role in several lymphoproliferative disorders (LPDs) occurring in immunocompromised and immunocompetent hosts.5⇓-7 Given the varied and nonspecific clinical manifestations, EBV antibodies and polymerase chain reaction (PCR) analysis in blood or CSF are often used for confirmation.8 The high soft-tissue contrast resolution of MR imaging is an essential tool during initial work-up and follow-up.9⇓⇓-12
Herein, we present an illustrative review of neuroimaging manifestations of EBV-associated disorders. Understanding the clinical context and awareness of the varied imaging characteristics can facilitate management of these potentially life-threatening conditions.
Pathogenesis
EBV is a double-stranded DNA human herpesvirus discovered in 1964. EBV is mainly transmitted through saliva; however, transmission through blood transfusions and organ transplants has been reported. EBV infects B lymphocytes, causing lytic and latent infections, and establishes life-long carrier status by residing in peripheral blood lymphocytes.13 EBV can invade the CNS through a hematogeneous route or retrograde along nerve fibers.14 The pathogenesis of EBV-associated neurologic disease is complex, resulting from direct infection, immunologic response, and reactivation of latent infection.4
EBV infection can potentially induce demyelination, neuroinflammation, and ADEM, leading to multifocal demyelination in the white matter of the brain. Latent EBV infection induces Epstein-Barr nuclear antigen (EBNA) expression that cross-reacts with myelin oligodendrocyte glycoprotein (MOG), producing anti-MOG antibodies.15⇓-17 The pathogenesis remains complex and poorly understood, but in many children with ADEM, elevated serum MOG levels are observed. Yet, no correlation has been observed between anti-EBNA-1 and anti-MOG immunoglobulin antibody titers in EBV-seropositive children.15 EBV-transformed B-cells may also secrete antineuronal or anti-EBV protein antibodies that cross-react with neuronal antigens.18 This likelihood is further compounded by a complex dysregulated immune response exacerbating CNS injury.14,19 EBV-specific B cells may enter the brain and form a site of antigen recognition with the help of other immune cells. A recent study found that EBNA-1 mimics the glial-cell adhesion molecule, a CNS protein expressed by myelin-forming cells. Antibodies targeting EBNA-1 cross-react with the glial-cell adhesion molecule in patients with MS, potentially causing autoimmune attacks on the CNS myelin.20
EBV is an oncogenic virus associated with various epithelial, mesenchymal, and hematologic malignancies. EBV-latent membrane protein (LMP1) genetic deletion and high EBNA-2 expression are important for EBV-positive primary CNS lymphomas (PCNSLs).21 In immunocompetent hosts, cytotoxic T-cells suppress EBV-infected lymphocytes, whereas immunosuppression leads to uncontrolled B-cell proliferation. The complex pathogenesis involves oncogenic transformation and tumor immune evasion and drives tumor cell proliferation, differentiation, invasion, metastasis, and resistance to apoptosis.17 The pathogenesis of EBV-related smooth-muscle cell tumors (EBV-SMTs) remains unclear. It is suggested that EBV infects SMTs through interaction with the CD21 receptor, similar to B lymphocytes. Other potential mechanisms include overactivation of the MTOR/Akt pathway and overexpression of the MYC proto-oncogene.22 The process of EBV infection and the pathogenesis of EBV-related diseases are briefly illustrated in the Online Supplemental Data.
Diagnosis of EBV CNS Infections
Given the nonspecific clinical symptomatology, the differential considerations include infectious, noninfectious inflammatory, metabolic, autoimmune, and neoplastic etiologies.23 Lesion distribution on MR imaging, duration of illness, immune status, and CSF analysis help narrow the differential considerations. Positive serum viral capsid antigen immunoglobulin (Ig) M or detection of EBV DNA through a PCR in CSF or serum or both are generally confirmatory.24
EBV Encephalitis
EBV is a rare cause of encephalitis in children and immuno-competent adults. The most common presentations are fever (81%), headache (66%), and seizure (10%).11 MR imaging is preferred for suspected encephalitis, with abnormalities found in 27.6%–80% of patients (Fig 1). Vyas et al12 reported abnormal findings on brain imaging in 29/45 (64.5%) pediatric patients. Cortical/subcortical (20%), white matter (15.5%), and basal ganglia (11%) involvement were more common, followed by the thalamus (9%), brainstem (6.2%), substantia nigra (4.4%), cerebellum (4.4%), and spinal cord (3%). Diffusion restriction and susceptibility changes were seen in 24.4% and 15.5% of cases, respectively. Meningeal involvement was reported in 5%–22%.9,12 Brain abscess, subdural effusion/empyema, and hemorrhage are rare.12
EBV meningoencephalitis in a patient with infectious mononucleosis. CSF was positive for EBV. Axial T2 FLAIR (A and B) images show multifocal areas of hyperintensity mainly within the subcortical white matter of the bilateral cerebral hemispheres (arrows) without diffusion abnormalities on axial DWI (C). Postcontrast axial T1-weighted image (D) shows diffuse leptomeningeal enhancement. The patient was fully recovered on follow-up.
Most patients fully recover without sequelae, while 5%–10% of cases may be fatal.4,11,25,26 For unclear reasons, patients with isolated hemispheric involvement generally have better prognoses. Similarly, about one-half of the patients with thalamic involvement have persistent deficits, while isolated brainstem involvement has a high mortality rate.9,11 EBV encephalitis in immunocompromised patients may occur secondary to latent virus reactivation or a primary infection after transplantation.27,28 The clinical and imaging manifestations are broadly similar, though cases with hemorrhage, ring enhancement, and masslike lesions may also be seen.27⇓-29 Correction of underlying immunosuppression is the mainstay of therapy.
EBV Cerebellitis
EBV-associated cerebellitis is rare and may occur secondary to infection or postinfectious inflammation from antineural antibodies.30⇓⇓-33 Frequent detection of EBV IgG and IgM antibodies in the serum and negative EBV CSF serology indicate a potential postinfectious or autoimmune phenomenon.29⇓-31 A positive CSF PCR suggests direct infiltration of the cerebellum by EBV, supported by evidence of EBV-encoded RNA expression in infiltrating inflammatory cells from cerebellar biopsies.32 Children are commonly affected; the condition often occurs following infectious mononucleosis. Studies have shown no MR imaging abnormalities32 to cerebellum swelling causing obstructive hydrocephalus.34 EBV-linked unilateral cerebellitis has been reported in immunocompromised adults with HIV.33 EBV cerebellitis is generally self-limiting, but IV immunoglobulin may reduce the severity and duration.31
EBV-associated cranial neuropathies are rare and generally manifest as facial nerve mononeuritis, though multiple cranial nerves may rarely be affected.35 Occasionally, oculomotor nerve palsy may manifest as the presenting symptom of primary EBV infection, with imaging demonstrating focal edema and enhancement at the root exit zone of the affected nerve (shooting star sign).36
Spinal Cord Involvement
ATM is generally idiopathic or postinfectious immune-mediated demyelination, with EBV being a rare culprit.4,37,38 EBV myelitis is characterized by T2 hyperintensity in the spinal cord that extends over ≥2 segments covering equal or more than two-thirds of the cord, with the thoracic cord being the most affected (Figs 2 and 3). Enhancement patterns range from none to diffuse, with leptomeningeal and nerve root enhancement reported.37,39,40 EBV serologies can aid in diagnosing postinfectious EBV myelitis/ATM because MR imaging findings resemble those of other ATM etiologies. Spinal cord ischemia, MS, neuromyelitis optica, and autoimmune-related myelitis are imaging differential diagnoses for ATM. MS plaques usually affect less than one-half of the cord area and may have concomitant brain lesions. Spinal cord infarction shows bilateral anterior horn cell T2 hyperintensity symmetrically.38 Although rare in immunocompetent individuals, EBV ATM should be considered in cases of high CSF cell count atypical for idiopathic ATM.40
EBV-related transverse myelitis, EBV IgM–positive. T2-weighted sagittal image of the thoracic spine (A) and axial images (B and C) reveal diffuse long-segmental hyperintense signal (arrows) in the spinal cord more in the central gray matter region along with mild cervical cord swelling. No abnormal enhancement on the sagittal T1-weighted postcontrast image (D) was seen.
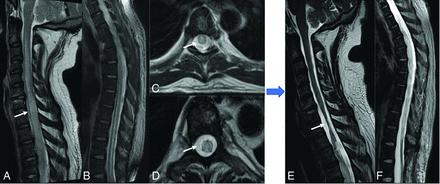
Presumed EBV myelitis, CSF EBV-positive. Conus biopsy revealed fibrin with macrophages, scant peripheral nerve myelins, and degenerating myelin, most consistent with infection. T2-weighted sagittal image of the cervical (A) and thoracic (B) spine along with axial images (C and D) reveal diffuse long-segmental T2-hyperintense signal in the spinal cord (arrows). Eight-month follow-up sagittal T2 (E and F) images show cord atrophy with persistent signal abnormality (arrow).
EBV-Associated Demyelinating Disorders
ADEM primarily affects children, with an incidence of 0.3–0.6/100,000, and may follow a viral infection or vaccination.41⇓-43 MR imaging typically shows asymmetric, multifocal white matter T2-hyperintense lesions. One-third of patients show ringlike or patchy peripheral contrast enhancement and restricted diffusion. Spinal cord involvement also occurs in about one-third of patients, manifesting as large lesions extending over multiple segments accompanied by cord edema.44
Differential considerations include MS, neuromyelitis optica, and acute hemorrhagic leukoencephalitis. Evidence of recent EBV infection, a monophasic course, and specific imaging features like periventricular sparing and the absence of Dawson fingers can help to differentiate the entity from MS.45 Patients usually respond to systemic steroids, and follow-up MR imaging typically shows resolution.46
Lymphoproliferative Disorders
Age-related impaired cell-mediated immunity or iatrogenic immune suppression increases the risk of several B-cell LPDs. These include lymphomatoid granulomatosis (LG), posttransplant and other immunodeficiency-associated LPDs, and B- and T-cell lymphomas.6,47,48
Lymphomatoid Granulomatosis
Under the World Health Organization Classification of Hematolymphoid Tumors (2022), LG is an LPD occurring exclusively in immunocompetent patients. Brain involvement is reported in up to 30% of patients with LG and is often secondary, with sporadic primary CNS involvement.49 Brain lesions may be diffusely infiltrating or masslike and show nodular, perivascular, or ringlike enhancement. Subependymal extension or leptomeningeal involvement may be present (Fig 4). LG typically exhibits an angiocentric and angiodestructive infiltrate consisting of lymphocytes, histiocytes, and sporadic plasma cells, with rare neutrophils and eosinophils. Necrosis may be observed in higher-grade lesions, while well-formed granulomas are not seen. The differential diagnosis for LG includes CNS lymphoma and vasculitis.50,51 Imaging findings are nonspecific and overlap with multiple tumoral and nontumoral pathologies, including glioblastoma, vasculitis, and chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS).50⇓⇓⇓-54 Treatment options include observation, resection, chemoradiation, corticosteroids, interferon, immunoglobulin, and rituximab.54
EBV lymphomatoid granulomatosis. A 40-year-old man with a history of HIV. EBV lymphomatoid granulomatosis was the finding of a prior right thalamus biopsy. Lymphohistiocytic infiltrate was the finding of a biopsy, consistent with EBV-associated lymphoproliferative disorder. Coronal T1-weighted postcontrast images show linear and punctate enhancement foci in the left parieto-occipital lobes (arrow).
EBV-Positive CNS Lymphoma
PCNSL is rare and, by definition, is confined to the brain, spinal cord, eyes, and CSF without systemic involvement. It is generally EBV-negative in immunocompetent patients and has a worse prognosis when EBV-positive.55 Immunodeficiency-associated PCNSLs account for 8%–10% of cases and corresponds explicitly to EBV-positive diffuse large B-cell lymphoma.6,56,57
PCNSL in immunocompetent patients may present as solitary or multifocal lesions that restrict diffusion and enhance homogeneously, often involving the white matter, corpus callosum, or basal ganglia (Fig 5).58 Compared with glioblastoma multiforme and metastases, PCNSLs show a lower CBV, peak height on the perfusion curve, and increased diffusion restriction due to cellular density.59 Stereotactic biopsy with histopathologic examination remains the criterion standard for diagnosis.60 Lee et al61compared MR imaging features in 10 EBV-positive PCNSLs with 45 EBV-negative PCNSLs (neither group with HIV/AIDS). Tumor necrosis, hemorrhage, and irregular or ring enhancement were common in EBV-positive PCNSLs (P < .001 each). However, ADC, maximum relative CBV, relative CBV, and Cho/NAA ratios were similar between the groups.
EBV-positive PCNSL in a 65-year-old male patient. T2-weighted (A) and FLAIR (B) images demonstrate the left parietal lobe necrotic T2-hyperintense lesion with a peripheral iso- to hypointense rim and surrounding white matter edema (arrows). The lesion demonstrates a hypointense rim (hemorrhage) (arrows) on the susceptibility-weighted (C) image, increased restriction on DWI (D, arrow), and a peripheral rim-enhancement pattern (arrows) on the postcontrast axial T1-weighted image (E).
PCNSLs in immunosuppressed patients are usually deep, periventricular, and often multiple, with necrosis, hemorrhage, and rim enhancement.6,55 Glioblastoma, metastases, and opportunistic infections are the primary differential.57,62⇓⇓-65
PTLD
PTLD is a rare EBV-driven polyclonal B-cell proliferation occurring in patients posttransplantation. CNS involvement is seen in 5%–20% of PTLDs after solid organ transplantation and portends a poor prognosis. Biopsy may be essential because in some cases, there is no EBV DNA in the plasma/CSF.49 Balaguer-Rosello et al identified 25 cases of EBV PTLDs in 1009 adult patients with allogeneic stem cell transplants and noted isolated CNS involvement in 36% of cases. Detection of plasma EBV DNA was significantly lower with isolated CNS PTLDs (33%) compared with extra-CNS disease (100%). MR imaging findings are nonspecific and overlap with other tumoral and nontumoral pathologies. These include solitary or multiple necrotic lesions with rim enhancement; perivascular, cranial nerve, or dural enhancement; and, rarely, choroid plexus involvement. The treatment involves reducing immunosuppression, chemoradiation, immunotherapy, and surgery.66
EBV-Related SMTs
EBV-SMTs of the CNS are rare mesenchymal tumors that develop predominantly in immunocompromised patients, with only a few cases reported in immunocompetent patients.67⇓⇓⇓⇓⇓-73 Lee et al67 were the first to link EBV with SMTs in immunocompromised patients. Symptoms depend on location, with some patients being asymptomatic. Diagnosis often requires a biopsy. Intracranial posttransplant SMT is typically along the dural sinuses, cerebral veins, and dura mater.68 Paez-Nova et al69 analyzed 47 cases of intracranial EBV-SMTs, with 40% being leiomyosarcomas and 60% being leiomyomas. EBV-SMTs resemble meningiomas on imaging and should be considered in HIV-positive and posttransplant patients of any age.70 Cavernous sinus involvement may occur and may present clinically as Tolosa-Hunt syndrome, but a negative response to steroids should suggest an alternate etiology, including EBV-associated neoplasia in immunosuppressed patients.68 EBV-SMTs appear as dural-based extra-axial lesions with iso- to hyperdensity and substantial enhancement on CT scans. On MR imaging, lesions are often hypo- to isointense on T1-weighted images, with T2 prolongation and variable enhancement (Fig 6). Unlike PCNSL, diffusion restriction is typically absent in EBV-SMTs.71 Spinal EBV-SMTs appear as intradural-extramedullary lesions and may resemble neurofibromas and schwannomas, with dumbbell-shaped tumors at the neural foramina.71,72 Schwannomas stain strongly for S100 protein, while EBV-associated SMTs show minor expression. In patients with HIV, immunohistochemistry can help to differentiate EBV-SMTs from Kaposi sarcoma, schwannoma, follicular dendritic cell sarcomas, and mycobacterial pseudotumor. In a study with 61 SMTs, desmin, smooth-muscle actin, muscle-specific actin, and CD21 were positive. Negative staining included S100 (27 cases), CD34 (17 cases), epithelial membrane antigen (15 cases), and cytokeratin (5 cases).73 EBV-SMTs exhibit short interlacing fascicles of atypical spindle cells with abundant eosinophilic cytoplasm.71,73 The optimal treatment for EBV-SMTs is unclear and may involve excision, radiation therapy, and/or chemotherapy. Although complete resection is the most effective treatment, 40% of patients undergo partial resection due to high risk.69 Recent studies have shown promise in alternative therapies such as mTOR/AKT inhibitors (sirolimus), EBV-specific immunotherapy, and demethylating agents.69,70
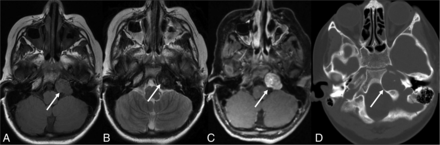
Biopsy-proved EBV-SMT. Axial FLAIR (A) and T2-weighted (B) images show a left petrous apex T2-hypointense lesion with central hyperintense foci and intense heterogeneous enhancement (arrows) on the postcontrast T1-weighted image (C) with bony remodeling (arrows) on the axial CT (D) image.
CONCLUSIONS
EBV in the CNS may be associated with a spectrum of infectious, noninfectious inflammatory, and lymphoproliferative disorders. MR imaging is preferred for evaluating and monitoring EBV-related CNS complications. Increased awareness of these uncommon but clinically relevant entities and their imaging spectrum may help with early diagnosis and therapy.
Footnotes
↵# Neetu Soni and Manish Ora are co-first authors.
Disclosure forms provided by the authors are available with the full text and PDF of this article at www.ajnr.org.
Indicates open access to non-subscribers at www.ajnr.org
References
- Received March 28, 2023.
- Accepted after revision May 26, 2023.
- © 2023 by American Journal of Neuroradiology